Amino Acids are wondrous things. Once you learn what they do in the body, you're almost struck with amazement. They fulfill the basic foundations of well-being like.Amino Acid Synthesis and Metabolism. However, the liver is the. In times of dietary surplus, the potentially toxic nitrogen of amino. In this respect. amino acids fall into three categories: glucogenic, ketogenic, or glucogenic. Glucogenic amino. TCA cycle intermediates, such as 2- oxoglutarate (. All amino acids except lysine and. Lysine and leucine are the only amino. Co. A or. acetoacetyl- Co. A, neither of which can bring about net glucose production. A small group of amino acids comprised of isoleucine. Finally, it should be recognized that amino acids have a third. During times of starvation the reduced carbon skeleton is used. CO2. and H2. O. Dietary protein digestion begins in the stomach via the actions of the pepsins, and continues. Within the small intestine there are two principal pancreatic enzymes. Several additional pancreatic. The initial enzyme involved in protein digestion is gastric pepsins. Get information, facts, and pictures about amino acid at Encyclopedia.com. Make research projects and school reports about amino acid easy with credible articles from. L-arginine is an amino acid that is the precursor to nitric oxide, a natural compound that helps relax your blood vessels and maintain normal blood pressure. The sulfur-containing amino acids (cysteine and methionine) are generally considered to be nonpolar and hydrophobic. In fact, methionine is one of the most. Pepsins are derived from the. Pepsins are released from pepsinogen via acid- induced autocatalysis. Pepsins hydrolyze peptide bonds. C- terminal side of aromatic and hydrophobic amino acids. Approximately 2. 0% of overall protein digestion is accomplished via the. Due to the acidic p.      
H optimum for the action of pepsins, these enzymes are inhibited. The remainder of protein digestion occurs within the duodenum and jejunum of the small intestine. Enteropeptidase is an enzyme secreted by. Lieberk. Following digestion, free amino acids, as well as. Although there is. The absorption of most amino acids from the intestine requires an active transport process that is dependent upon. Na+ or H+ co- transport. These three broad categories are the neutral. All of these transporters are members of the. SLC) family of transporters that includes the SLC1, SLC6, SLC7, SLC3. SLC3. 8, and SLC4. The transport of amino acids occurs in all other cells and so these transport systems are expressed throught the body. They are critically important within the context of the brain and the kidney. Many of the transporters that are required for intestinal amino acid uptake are also expressed in the renal tubules where they are responsible for excretion and reabsorption of amino acids. The neutral amino acid transporters were originally subdivided into at least eight subfamilies with all but one being Na+- dependent transporters. These original designations were the system A, ASC, N, beta (. 
The system A and system B0 transporters were those defined by their preference for alanine and other small and polar neutral amino acids. The system ASC transporters were defined by their preference for alanine, serine, and cysteine. The system L transporters, which are Na+- independent, were defined by their preference for leucine and other. The system N transporters show preference for glutamine, asparagine, and histine and are restricted to expression within hepatocytes. The acidic amino acid transporters were also subdivided into several subfamilies, all of which are Na+- dependent transporters. The acidic transporter subfamilies were the system B0,+, b+, y+, y+L, and b. The anionic amino acid transporters were also referred to as the system X transporters. The XAG– transporters are potassium- dependent glutamate and aspartate transporters. The XAG– transporters are now more commonly called the excitatory amino acid transporters (EAAT) with there being five members EAAT1 (SLC1. A3), EAAT2 (SLC1. 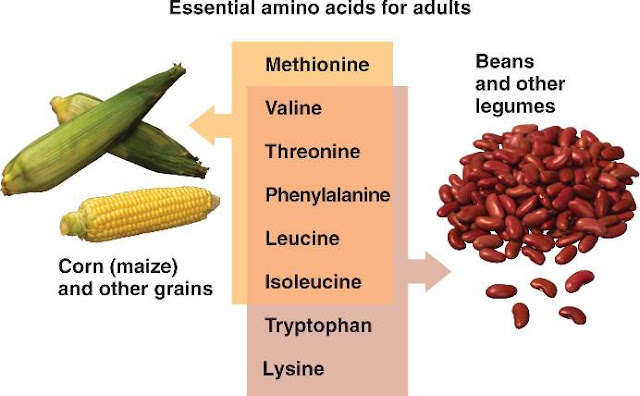
A2), EAAT3, (SLC1. A1), EAAT4 (SLC1. A6), and EAAT5 (SLC1. 
A7). The Xc– transporters are cystine and glutamate transporters that transport cystine in the opposite direction to that of glutamate (antiporter). Due to the inconsistent usage of the varied nomenclature systems for amino acid transporters a nomenclature system encompassing five transport activities has been proposed. These five systems designations are: 1) the neutral system (or methionine preferring system) which transports all neutral amino acids; 2) the basic system which is responsible for the transport of cationic amino acids in addition to cystine; 3) the acidic system which transports glutamate and aspartate; 4) the iminoglycine system which transports proline, hydroxyproline, and glycine; and 5) the . Within intestinal enterocytes the absorbed peptides are. The free amino acids are then transported. Essential vs. Nonessential Amino Acids. Nonessential. Alanine, Asparagine, Aspartate, Cysteine, Glutamate, Glutamine, Glycine. Proline, Serine, Tyrosine. Essential. Arginine*, Histidine, Isoleucine, Leucine, Lysine, Methionine*. Phenylalanine*, Threonine, Tryptophan, Valine*The amino acids arginine, methionine and phenylalanine are. Methionine is required in large amounts to produce. Humans express two different AST enzymes, both of which function as homodimeric enzymes. One AST enzyme is a cytosolic enzyme and the other is a mitochondrial enzyme. The cytosolic AST enzyme is synthesized by the GOT1 gene (glutamate- oxalate transaminase 1) that is located on chromosome 1. The mitochondrial AST enzyme is synthesized from the GOT2 gene that is located on chromosome 1. RNAs that encode two different isoforms: isoform 1 (4. Reaction catalyzed by ASTAspartate can also be derived from asparagine (whose synthesis is outlined. Quite often, we receive calls from families who are distraught and worried after trying five or six different infant formulas. The importance of aspartate as a precursor of ornithine for the urea cycle is. Nitrogen Metabolism page. In addition, the use of asparaginase as an anti- cancer therapeutic, for certain types of leukemia, stems from the fact that, although. These cells acquire the needed exogenous asparagine from the plasma. Chemotherapy using the. The asparaginase gene (symbol: ASPG) is located on chromosome 1. Reaction catalyzed by asparaginaseback to the top. Alanine and the Glucose- Alanine Cycle. Aside from its role in protein synthesis, alanine is. In this. capacity it serves a unique role in the transfer of nitrogen from peripheral. Alanine is transferred to the circulation by many tissues. Liver accumulates plasma. The pyruvate is either oxidized or converted to. When alanine. transfer from muscle to liver is coupled with glucose transport from liver back. Glucose oxidation produces pyruvate which can. This reaction is catalyzed by alanine. ALT (ALT used to be called serum glutamate- pyruvate. SGPT). Additionally, during periods of fasting, skeletal muscle. The alanine then enters the blood. Within the liver alanine is converted. The amino group transported from the muscle to the liver in the form. Cysteine Biosynthesis: Role of Methionine. The sulfur for cysteine synthesis comes from the essential. Initially a condensation of ATP and methionine, catalyzed by. MAT), yields S- adenosylmethionine (SAM or. Ado. Met). In the production of SAM all phosphates. ATP are lost: one as Pi and two as PPi. It is. adenosine which is transferred to methionine and not AMP. MAT is also called S- adenosylmethionine synthetase. There are three MAT genes in humans identified as MAT1. A, MAT2. A and MAT2. B. The MAT enzymes encoded by the MAT1. A gene function as either a homotetramer identified as MAT I (called the alpha form), or as a homodimer identifed as MAT III (called the beta form). The MAT I and MAT III isoforms are only expressed in the liver. The MAT enzyme encoded by the MAT2. A gene is identified as MAT II (called the gamma form). The MAT II enzyme is expressed in several non- hepatic tissues. The MAT1. A gene is located on chromosome 1. The MAT2. A gene is located on chromosome 2p. The MAT2. B gene is located on chromosome 5q. RNAs encoding the MAT 2 beta isoform 1 (3. MAT 2 beta isoform 2 (3. Biosynthesis of S- adenosylmethionine, SAMSAM serves as a precurosor for several methyl. Specialized Products of Amino Acids). Transmethylation reactions employing SAM are. S- adenosylmethionine in. S- adenosylhomocysteine is then cleaved by adenosylhomocyteinase (also called S- adenosylhomocysteine hydrolase) to yield homocysteine and adenosine. Adenosylhomocysteinase is encoded by the AHCY gene located on chromosome 2. RNAs encoding adenosylhomocysteinase isoform 1 (4. Homocysteine can be converted back to methionine by methionine synthase (also called homocysteine methyltransferase). This reaction was also discussed in the context of vitamin B1. Vitamins page. The methionine synthase reaction represents the clinically most significant of only two vitamin B1. Methionine synthase, in addition to B1. N5- methyltetrahydrofolate (N5- methyl THF). Methionine synthase is encoded by the MTR gene (5- methyltetrahydrofolate- homocysteine methyltransferase) located on chromosome 1q. RNAs. Deficiency in B1. MTR gene all will result in the development of. The anemia develops due to the trapping of folate in the reduced form (N5- methyl- THF) which results in depletion of the active folate pool. The active folate pool is required for purine and thymine nucleotide biosynthesis needed for DNA synthesis. Bone marrow erythroblasts begin to enlarge in preparation for cell division but, due to loss of nucleotide synthesis, they remain as megaloblasts. In cysteine synthesis, homocysteine condenses with. The details of CBS are discussed below. Cystathionine is subsequently cleaved by cystathionine. The sum of the. latter two reactions is known as trans- sulfuration. Cysteine is used for protein synthesis and other body. While cysteine readily oxidizes to form the. Utilization of methionine in the synthesis of. The sulfur from the essential amino acid, methionine, is required for the synthesis of cysteine. The synthesis of cysteine represents an extremely important and clinically relevant biochemical pathway. Several vitamins are required for this metabolic pathway to proceed emphasizing the nutritional impact. Folate, pyridoxal phosphate (PLP, B6), and B1. The enzyme methionine synthase requires both folate and B1. Deficiency in either of these vitamins contributes to homocysteinemia and also to the development of macrocytic (megaloblastic) anemias. Cystathionine . Both of these enzymes require pyridoxal phosphate (PLP: derived from. B6) as a. cofactor, and both are under regulatory control. Cystathionine . In addition. Foods Containing the Amino Acid Lysine. Your body can't make the amino acid lysine, so you need to get it through your diet. Some people, including those suffering from herpes, may want to consume extra lysine. Reducing your intake of the amino acid arginine and consuming more lysine can help minimize herpes outbreaks, according to an article published in . The typical dosage for preventing or treating herpes is 3,0. University of Maryland Medical Center. Pork is high in lysine. Photo Credit Alex. Pro. 95. 00/i. Stock/Getty Images. Foods containing lysine include pork, beef, cod, sardines, poultry, eggs, dairy products, lentils, soy beans, brewer's yeast and nuts. A 3- ounce serving of top- round roast provides 3,0. A 3- ounce serving of rotisserie chicken has 2,5. Vegetarian foods tend to be lower in lysine, with boiled soybeans providing 1,9.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2017
Categories |
 RSS Feed
RSS Feed
